 Knowing this value, we can adjust the temperature to drive the process to spontaneity or alternatively to prevent the process from occurring spontaneously. Sometimes it can be helpful to determine the temperature when Δ G° = 0 and the process is at equilibrium. Campfire, Ice melting, salt or sugar dissolving, popcorn making, and boiling water are some entropy examples in your kitchen. If Δ H is positive, and – TΔ S negative, the reaction will be spontaneous at high temperatures (increasing the magnitude of the entropy term). Entropy measures how much thermal energy or heat per temperature. If Δ H is negative, and – TΔ S positive, the reaction will be spontaneous at low temperatures (decreasing the magnitude of the entropy term). As shown in Table 18.2 “Spontaneity and the Signs of Enthalpy and Entropy Terms,” the temperature can be the deciding factor in spontaneity when the enthalpy and entropy terms have opposite signs. There is a constant amount of energy in the universe, but the way it is distributed is always changing. It is proved that the relative entropy for a quantum system is non-increasing under a trace-preserving completely positive map. Since all temperature values are positive in the Kelvin scale, the temperature affects the magnitude of the entropy term. Entropy is not energy entropy is how the energy in the universe is distributed. Table 18.2 Spontaneity and the Signs of Enthalpy and Entropy Terms Δ H 
Depending on the sign and magnitude of each, the sum of these terms determines the sign of Δ G and therefore the spontaneity (Table 18.2 “Spontaneity and the Signs of Enthalpy and Entropy Terms”). If we examine the Gibbs free energy change equation, we can cluster the components to create two general terms, an enthalpy term, Δ H, and an entropy term, – TΔ S. Transcribed image text: QUESTION 17 Which figure represents a process with a positive entropy change O O 8 808 80 18 00 008 98 00 8 8. The temperature plays an important role in determining the Gibbs free energy and spontaneity of a reaction. A minimal dynamical system which has completely positive entropy, and does not have uniformly positive entropy is constructed. We have seen how we can calculate the standard change in Gibbs free energy, Δ G°, but not all reactions we are interested in occur at exactly 298 K. But like enthalpy, changes in entropy alone cannot. 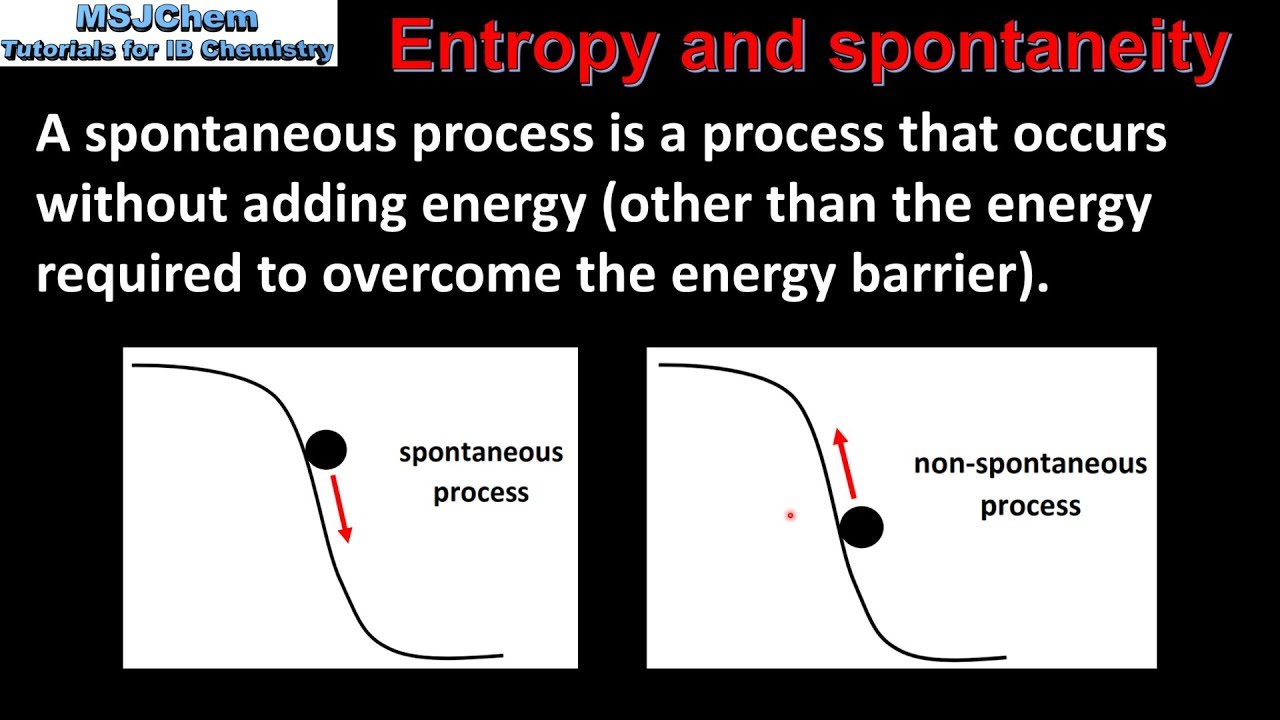
In the Gibbs free energy change equation, the only part we as scientists can control is the temperature. When a change entropy is positive, it makes the change more spontaneous (favorable). Entropy can have a positive or negative value. It is denoted by the letter S and has units of joules per kelvin. 
The value of entropy depends on the mass of a system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
